Water is the main nutrient in animal feed, it has a direct impact on biological cycles, the absorption of nutrients and is decisive for maintaining adequate physiological constants. Water is also the major component of the animal body, representing between 70% and 80% of the total. The physicochemical and microbiological characteristics of our water will affect the normal development of our farms.
 In this article, we are going to talk about two physical and chemical parameters that significantly affect water quality: HARDNESS. Although they are often related, they are two independent characteristics that must be treated individually. These two parameters can cause some health problems and can affect the biocidal treatment we do in the water. In some way, they can influence a good purification and an adequate distribution of water.
In this article, we are going to talk about two physical and chemical parameters that significantly affect water quality: HARDNESS. Although they are often related, they are two independent characteristics that must be treated individually. These two parameters can cause some health problems and can affect the biocidal treatment we do in the water. In some way, they can influence a good purification and an adequate distribution of water.
The hardness of the water.
Hardness is the sum of mineral salts dissolved in water: calcium (Ca ++) and magnesium (Mg ++) represented in equivalent amounts of calcium carbonate, carbonates, bicarbonates and sulphates. Calcium and magnesium salts. Water, except for distilled water, contains dissolved calcium and magnesium carbonate salts. The concentration of these salts determines the hardness of the water, which can be expressed as calcium carbonate or magnesium carbonate. The sum of these two represents the TOTAL HARDNESS level.
The presence of dissolved salts is because the water often comes from underground sources, which are formed from filtered rain in the deep layers of the earth. The presence of hard water in our farms is a very common reality, for this, we need to know how to identify the hardness, to know how it affects us and to know what treatments are recommended to face this type of water.
Types of HARDNESS:
- Temporary Hardness: It is determined by the content of carbonates and bicarbonates calcium and magnesium. It can be eliminated by boiling the water and subsequent elimination of precipitates formed by filtration, it is also known as “Carbonate Hardness”
- Permanent Hardness: it is determined by all calcium and magnesium salts except carbonates and bicarbonates. It is also known as “Non-carbonate hardness”. Permanent hardness cannot be removed by boiling water.
Measurement of hardness:
The hardness of water can be expressed in different ways. The French degree, all the components are expressed as CaCO3, and in this case, 10 mg / l of CaCO3 is equivalent to 1 French degree. Another way to express hardness is in parts per million (ppm = mg / l), of calcium carbonate or calcium oxide, which is the most common form used in Spain and Latin American countries.
The equivalences between the different units are given below:
- 1.25 English degrees = 1.78 French degrees = 1.04 American degrees = 17.9 ppm (17.9 mg / l) of calcium carbonate = 10 ppm (10 mg / l) of calcium oxide.
- 1 French degree (ºHF) = 4 mg / L Ca or 2.43 mg / L Mg or 10 mg / L CaCO3
It can also be measured based on the mg / L of Calcium Carbonate, this being the most common measurement. This data is calculated by an equivalence of the concentration of Ca ++ and Mg ++ salts.
- mg / l Ca x 2.5 + mg / l Mg x 4.2) / 10
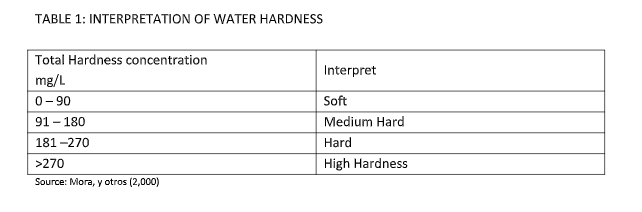
In order to interpret the hardness, the type of water hardness is shown in the table. According to their concentration.
 What consequences does hardness have on our farms?
What consequences does hardness have on our farms?
- Direct consequences for animal health.
Although the hardness of the water does not pose a serious disorder for animals, high concentrations of total hardness in drinking water can interfere with the intestinal absorption of microelements, they can reduce the effectiveness of certain chelation drugs (Oxytetracycline) In addition, our animals They have homeostatic mechanisms that allow regulating mineralization in the blood, we must remember that Calcium is a fundamental mineral in the rapid development that our animals have in fattening phases.
In laying hens, extreme hardness levels, above 500 mg of calcium carbonate, in drinking water, limit the absorption of calcium in the intestine, essential for the formation of the eggshell

- Technical consequences on equipment and facilities.
The most significant technical consequences of having hard water on our farms will be the problems derived from incrustations in pipes and drinking fountains, which reduce the flow of water. The diameter of the pipes will be reduced, reducing the water pressure and therefore the availability of water for the animals, in addition, it is capable of blocking nipples and teats, which can cause serious dehydration problems. One of the most common problems on our farms is the calcification of the evaporative panels of the farms for cooling. This reduces their lifespan and generates heat stress for the animals.
The mineral salts of calcium and magnesium do not have a direct relationship with the biofilm, but it is an indirect biofilm development factor since the roughness caused by the precipitation of these salts on the water pipes and drinkers makes it easier for the adhesion of the biofilm and protection of microorganisms. Calcium always precipitates more when the water velocity is lower, for example, there will be a greater possibility of calcium precipitating in the first week of the life of the chicks, due to the low water consumption. In addition, at this time the temperature can reach 30ºC, and the higher the water temperature, there is usually more precipitation. Calcium can cause problems in all water misting systems, since it blocks the mists, both in systems for supplying humidity within the farm, as well as supplying humidity in hatchers.
- Technical consequences in the use of detergent and disinfectant products
Detergents, in hard water conditions, generate worse foam, forcing us to use higher doses of detergent to obtain the same foam quality. If the water is very hard, you usually have to increase the dose of detergents. If the water is very hard, some disinfectants are less stable in dilution, especially due to the coincidence of hard water with a high pH.
- Softening
There are three basic ways to soften water:
- Force the precipitation of insoluble substances, such as calcium carbonate CaCO3 and magnesium hydroxide Mg (OH) 2, before they enter the water system.
- Ion exchange resins which consist of removing calcium Ca2 + and magnesium Mg2 + ions from water.
- Prevent calcium ions (Ca2 +) from calcium carbonate CaCO3 from precipitating through complex formation.
- Over-softening.
Great care must be taken when using total softening methods in water. Excessive water softening can also have adverse effects on the corrosion of galvanized iron and steel pipes. Very soft water can also contribute to the wear of concrete tanks. For the drinking water of animals, total elimination of hardness is not recommended, it can cause intestinal problems of absorption of nutrients, causing soft feces.
 BBZIX RECOMMENDATIONS FOR HARD WATER ON AGRICULTURAL FARMS
BBZIX RECOMMENDATIONS FOR HARD WATER ON AGRICULTURAL FARMS
In a farm, we must always keep in mind what Physical Chemical characteristics our water has. Let us remember that it is the nutrient with the highest consumption and that is why we must have its Technical Data Sheet, which is given by a CF analysis. In these analyses, we will look at the Total Hardness and the Calcium Hardness and make decisions.
As long as the Total Hardness does not exceed 250 mg / l of CO3Ca, we do not recommend any treatment for drinking water. In most farms, we only have one source of water and it is used for everything. The systems that we have discussed in this article involve a high cost in both equipment and chemicals for optimal maintenance.
A hardness that exceeds 300 mg / l of CO3Ca and is causing us continuous problems of calcium deposition in pipes and/or cooling panels, it is advisable to install ion exchange resins but that only act in 50% of total water, placing a bypass, thus only half of the water will be treated, resulting in water with half the initial Hardness.
Working with those Hardness levels of between 100-250 mg / l of CO3Ca, it is possible that there are calcium deposits in some points (pipes, drinkers, panels, etc). It is much cheaper to have well-established BBZIX cleaning and disinfection protocols.
PIPES DESCALING: With ZIX VIROX AND AZIX FORTE

ZIX VIROX
DESCALING OF REFRIGERATION PANELS: With ZIX VIROX, AQUAZIX PLUS AG, and AZIX FORTE.

ZIX VIROX

AQUAZIX PLUS AG

AZIX FORTE





